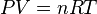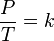Gas Laws
Gas Laws
The gas laws are a set of laws that describe the relationship between thermodynamic temperature (T), pressure (P) and volume (V) of gases. It is a loose collection of rules developed between the late Renaissance and early 19th century.
which with the addition of Avogadro’s law later gave way to the ideal gas law:
where
- P is the pressure in Pascal
- V is the volume in Cubic metres
- n is the number of moles of gas
- R is the ideal gas constant (8.31 J/mol K)
- T is the temperature in kelvins.
(The above units are the SI ones. The law works equally well with any consistent set of units, provided that the temperature scale is such that it is zero at absolute zero.)
Other important gas laws include Dalton’s law of partial pressures. The kinetic theory of gases, Graham’s law of effusionand root mean square velocity all explain how individual molecules in a gas act and their relation to pressure, volume, and temperature.
A gas that obeys these gas laws exactly is known as an ideal gas (or perfect gas). An ideal gas does not exist; however, some gases follow the laws more closely than others given standard conditions.
AVOGADRO’S LAW
Equal volumes of all gases at the same temperature and pressure contain the same number of molecules.
Avogadro’s law is one of the gas laws. The law is named after Amedeo Avogadro, who in 1811 hypothesized that equal volumes of gases, at the same temperature and pressure, contain the same number of particles, or molecules. Thus, the number of molecules in a specific volume of gas is independent of the size or mass of the gas molecules
The minor aspect of the law can be stated mathematically as:
.
-where:
- V is the volume of the gas.
- n is the number of moles in the gas.
- a is a constant.
However, this above equation is just a trivial one, which is valid for all homogeneous substances, including homogeneous liquids and solids. This relation is easy to deduce, its validity was assumed before Avogadro’s work.
The most important consequence of Avogadro’s law is the following: The ideal gas constant has the same value for all gases. This means that the constant const of
has the same value for all gases, independent of the size or mass of the gas molecules. This statement is nontrivial, and it embodies Avogadro’s ingenious insight in the nature of ideal gases. It took decades to prove Avogadro’s law based on the kinetic theory of gases.
One mole of an ideal gas occupies 22.4 liters (dm3) at STP. This is often referred to as the molar volume of an ideal gas. Real gases may deviate from this value.
The number of molecules in one mole is called Avogadro’s number: approximately 6.022×1023 particles per mole.
Avogadro’s law, together with the combined gas law, forms the ideal gas law.
Boyles Law
The volume of a given mass of gas at a constant temperature is inversely proportional to its pressure.
If the pressure of a gas is doubled, then its volume halved. In the equation form pV = constant, or
p1V1 = p2V2 where the subscripts 1 and 2 refer to the values of the pressure and volume at any two readings during the experiment.
Charles’s Law
Lussac’s Law
The other law, discovered in 1802, states that the pressure of a fixed amount of gas at fixed volume is directly proportional to its temperature in kelvins. It is expressed mathematically as:
-Where:
- P is the pressure of the gas.
- T is the temperature of the gas (measured in kelvins).
- k is a constant.
This law holds true because temperature is a measure of the average kinetic energy of a substance; as the kinetic energy of a gas increases, its particles collide with the container walls more rapidly, thereby exerting increased pressure.
For comparing the same substance under two different sets of conditions, the law can be written as:
Charles’s Law was also known as the Law of Charles and Gay-Lussac, because Charles used some of Gay-Lussac’s data to formulate his law. However, in recent years the term has fallen out of favor since Gay-Lussac has the second but related law presented here attributed to him. This related form of Gay-Lussac’s Law, Charles’s Law, and Boyle’s law form the combined gas law. The three gas laws in combination with Avogadro’s Law can be generalized by the ideal gas law.